Kelly, Resa M

Professor, Chemistry
Director, Science Education
ACS IAC Board Member (2020-2022)
Preferred: resa.kelly@sjsu.edu
Telephone
Preferred: (408) 924-4940
Office Hours
Arrange through email
Education
- Doctor of Philosophy, Univ of Northern Colorado, 2005
- Master of Science, Univ of Northern Colorado, 2004
- Master of Arts, Univ of Northern Iowa, 2000
- Bachelor of Arts (Chemistry), Univ of Northern Iowa, 1992
- Bachelor of Arts (Psychology), Univ of Notre Dame, 1990
Bio
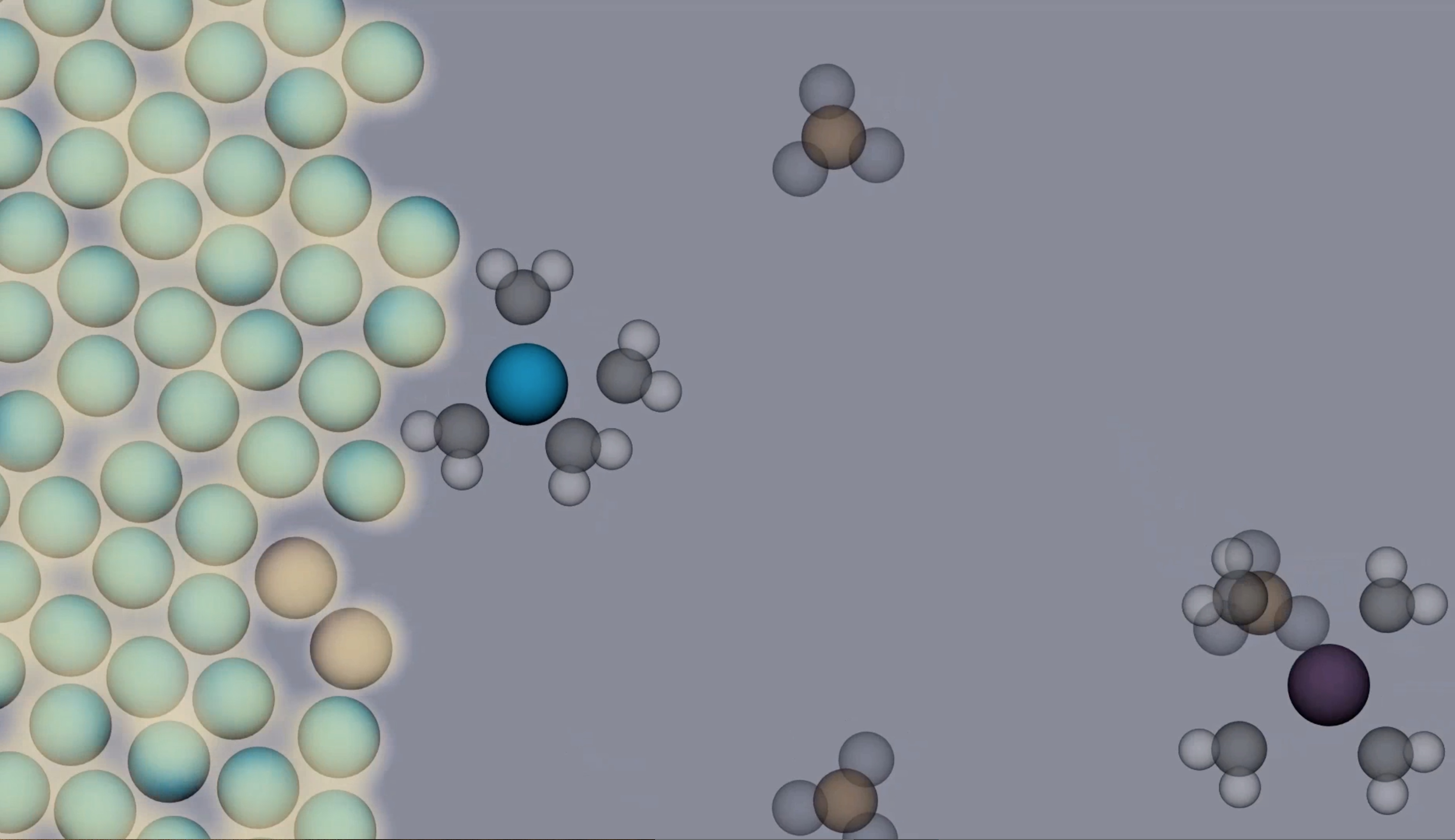
Professor Kelly's research interests involve studying how students learn and modify their understanding of chemistry concepts and mechanisms after viewing and reflecting on molecular visualizations. She also explores best practices for the design of molecular visualization tools (see the link below -Design Principles for Effective Molecular Animations). Dr. Kelly enjoys hiking and fitness classes in her free time.
General Chemistry Videos and Animations
Videos
Animations
Chemical Education Research (CER)
In the Kelly group, we study the teaching and learning of chemistry across secondary and tertiary levels. We are particularly interested in exploring ways to get students to critique molecular level visualizations in connection to experimental evidence.
Current research focuses on:
- Designing visualizations of molecular level events and mechanisms (submicroscopic emphasis)
- Developing videos of experimental evidence (macroscopic emphasis) complete with experimental controls and variables
- Exploring the use of metacognitive monitoring activities partnered with visualizations
- Performing mental model analysis through picture construction, think aloud and semi-structured interviews
Current Graduate Students:
Justin Choi - Exploring how students make use of "cheat sheets" for taking chemistry exams.
Priya Srinath - Examining ways that students engage with sensemaking when modeling the atomic level and when reviewing conflicting animations.
Current Undergraduate Students:
Accepting students, please contact Professor Kelly
Former Graduate Students:
Brandon Wall - MA Science Education 2018 - Project title: Analyzing how students apply their understanding of atomic level animations of precipitation reactions to revise their atomic level model of a neutralization reactions
Melissa Harker - MA Science Education 2015 - Project title: The effect of participating in a simulated scientific community on seventh graders' conceptions of the nature of science
Cristina Chang - MA Science Education 2013 - Project title: A case study of teachers' understanding of sustainability
Juliet Hamak - MA Science Education 2013 - Project title: Analysis of general chemistry students' understanding of electrolytic solution characteristics
Interested in CER?
- SJSU undergraduates should schedule an appointment during office hours.
- Prospective graduate students should contact Dr. Kelly (#4-4940 or email)
Publications
Kelly, R. M.; Akaygun, S.; Hansen, S. J. R.; Villalta-Cerdas, A; Adams, J. (2021). Examining learning of atomic level ideas about precipitation reactions with a resources framework. Chemistry Education Research and Practice, 2021, DOI: 10.1039/D0RP00071J
Kelly, R. & Akaygun, S. (2019) Visualizations and representations in chemistry education (Editorial). Chemistry Education Research and Practice, 20, 657-658.
Hansen, S. J. R.; Hu, B.; Riedlova, D.; Kelly, R. M.; Akaygun, S; Villalta-Cerdas, A. (2019). Critical consumption of chemistry visuals: eye tracking structured variation and visual feedback of redox and precipitation reactions. Chemical Education Research and Practice. DOI:10.1039/c9rp00015a
Herrington, D.;Sweeder, R.;Daubenmire, P.; Bauer, C.; Bretz, S.; Bunce, D.; Carmel, J.; Cole, R.; DeKorver, B.; Kelly, R.; Lewis, S.; Hoyo, M.; Ryan, S.; Stains, M.; Towns, M.; Yezierski, E. (2019). Supporting the growth and impact of the chemistry-education-research community. Journal of Chemical Education. DOI: 10.1021/acs.jchemed.8b00823.
Akaygun, S.; Adadan, E.; Kelly, R. (2018). Capturing preservice chemistry teachers’ visual representations of redox reactions through storyboards. Israel Journal of Chemistry.
Kelly, R. M. (2018). The Division of Chemical Education's International Activities Committee: Insights from Chairs - Past and Present. International Perspectives on Chemistry Education Research and Practice. Cox, C.; Schatzberg, W. Eds; American Chemcial Society: Washington, DC. bk-2018-00068x.R1.
Kelly, R. M.; Akaygun, S.; Hansen, S. J. R.; Villalta-Cerdas, A. (2017). The effect that comparing molecular animations of varying accuracy has on students' submicroscopic explanations. Chemical Education Research and Practice. DOI: 10.1039/C6RP00240D.
Kelly, R. M. (2017). Learning from contrasting molecular animations with a metacognitive monitoring activity. Educación Química. http://doi.org/10.1016/j.eq.2017.02.003
Kelly, R. M.; Hansen, S. J. R. (2017). Exploring the design and use of molecular animations that conflict for understanding chemical reactions. Química Nova.http://dx.doi.org/10.21577/0100-4042.20170043
Kelly, R. M.; Akaygun, S.(2016). Insights into how students learn the difference between a weak acid and a strong acid from cartoon tutorials employing visualizations. Journal of Chemical Education. doi:http://dx.doi.org/10.1021/acs.jchemed.6b00034.
Kelly, R. M. (2016). ConfChem Conference on Interactive Visualizations for Chemistry Teaching and Learning: Insights into Molecular Visualization Design. Journal of Chemical Education. doi: http://dx.doi.org/10.1021/acs.jchemed.5b00556.
Chang, H. C.; Kelly, R. M.; Metzger, E.P. (2016). A Qualitative Study of Teachers' Understanding of Sustainability: Education for Sustainable Development (ESD), Dimensions of Sustainabililty, Environmental Protection. Improving K-12 STEM Education Outcomes through Technological Integration (p.p.1-436); Urban, M. J.; Falvo, D. A. Eds.; Hershey, PA: IGI Global. do:10.4018/978-1-4666-9616-7.
L. L. Jones; Kelly, R. M. (2015). Chapter 8 -Visualization: The Key to Understanding Chemistry Concepts. Sputnik to Smartphones: A Half-Century of Chemistry Education (p.p. 121-140); Orna, M. V. Ed.; American Chemical Society: Washington, DC. doi: 10.1021/bk-2015-1208.ch008.
Kelly, R. M. (2015). Insights into visualization design. DivCHED CCCE 2015 Spring ConfChem. http://confchem.ccce.divched.org/2015SpringConfChemP1: May 8-14, 2015.
Kelly, R.M. (2014). Using variation theory with metacognitive monitoring to develop
insights into how students learn from molecular visualizations. Journal of Chemical Education 91(8), 1152-1161.
Kelly, R. (2013). How a qualitative study with chemistry instructors informed atomic level animation design. Pedagogic Roles of Animations and Simulations in Chemistry Courses; Suits, J.; Sanger, M., Eds.; American Chemical Society: Washington, DC.
Kelly, R. M., Barrera, J. H., & Mohamed, S. C. (2010). An Analysis of Undergraduate General Chemistry Students’ Explanations of the Submicroscopic Level of Precipitation Reactions. Journal of Chemical Education 87(1), 113-118. Publication date(Web): December 18, 2009.
Bishop, M. & Kelly, R. (2009). Bound to fail: challenges faced in the design of molecular level visualizations (Paper 6) Committee on Computers in Chemical Education Newsletter Publication date(Web): December 7-9, 2009.
Kelly, R. M. & Jones, L. L. (2008). Investigating students' ability to transfer ideas learned from molecular animations of the dissolution process. Journal of Chemical Education 85 (2), 303-309.
Kelly, R. M. & Jones, L. L. (2007). Exploring how different features of animations of sodium chloride dissolution affect students' explanations. Journal of Science Education and Technology 16(5), 413-429.
Kelly, R., Phelps, A., & Sanger, M. (2004). The effects of a computer animation on students' conceptual understanding of a can-crushing demonstration at the macroscopic, microscopic, and symbolic levels. Chemical Educator 9(3) 184-189.